 |
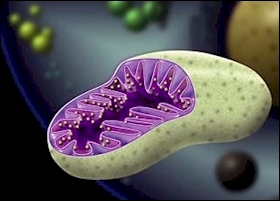
| Free radicals do the most damage when they attack a cell's mitochondrion, like this one. The mitochondria house DNA and free radical attacks can trigger mutations. |
The spokes and globes that shimmer and wheel slowly on Jim Wright's computer monitor resemble Tinker Toys but to him, they could be the Fountain of Youth.
Wright, a chemistry professor at Carleton University in Ottawa, is trying to design a molecule he hopes might someday be able to help slow down heart disease, cancer, arthritis and aging.
In November 2000, Wright and the five other members of the team received a three-year $413,360 grant from the Natural Sciences and Engineering Research Council (NSERC) to try to create what Wright describes as a "souped-up version" of Vitamin E and study its effects.
The goal is to create a molecule that can combat free radicals — unstable oxygen molecules that wreak havoc.
"Free radicals are like wolves and our bodies are made out of molecules which are like a herd of moose," Wright says.
"In that herd, there are some weak links — the very old, the crippled, the very young. That's us. That's our DNA, our proteins, our fats. The wolves pick off these weaker members."
Electron knights storm the castle
Every cell in the human body is powered by oxygen. Just as a car produces carbon dioxide when it burns gas, these cells give off "emissions" when they burn oxygen. The byproduct is oxidants, or free radicals — molecules that lack an electron. Normal human metabolism isn't the only way free radicals are created: they also spring from cigarette smoke, radiation, sunlight, air pollution and bad diet.
But no matter how they're created, all are highly volatile molecules that try to steal electrons wherever they can.
The quest for a
'souped-up' Vitamin E
The experiment has six stages:
• First, Wright uses a high-powered computer and software that allows him to design the molecules. Once he develops a formula he likes, he sends it to the other members of the team for feedback. Sometimes they tell him a molecule is too hard to make, too dangerous or too weak. Other times, they give him the thumbs up.
• Once approved, the molecule leaps the gap between the virtual and physical worlds — it's created in a laboratory by Tony Durst, a chemistry professor at the University of Ottawa.
• Then Keith Ingold, a scientist at the National Research Council in Ottawa, tests it in various lab solvents to see if it reacts against free radicals as expected. Ingold has been renowned world-wide since 1960s for his work on free radicals and Vitamin B.
• The fourth stage, testing the molecule on fake cells, is carried out by Ross Barclay at Mount Allison University in Sackville, N.B.
• Once that hurdle is leaped, David Miller, a cell culture expert and chemistry professor at Carleton University tests it on actual human leukemia cells.
• Finally, Carleton University neuroscientist Bruce Pappas will test the molecule on rats. After partially closing an artery close to the brain to induce stroke, he plans to feed the rats a compound containing the antioxidants. He will then measure the damage to the brains through a series of tests, including autopsies. |
Healthy cells can normally fight off these electron bandits. But when cells are damaged or become less efficient as the body ages, free radicals can wreak havoc.
Picture knights storming a castle. In their insatiable quest for oxygen, the free radicals will attack virtually any part of the cell. They pillage electrons from molecules in the cell walls, for example, setting off chain reactions that either make the walls too stiff to do their jobs or so weak that the innards leak out.
But the most ominous destruction occurs within the mitochondria, the cell's power
plants, which break down fats, carbohydrates and proteins to
produce energy. In the process, free radicals are
created.
The mitochondria house a
single DNA ring molecule. When free radicals attack the
ring, they can trigger mutations.
Just as exposure to oxygen browns apples, rusts metals and turns butter rancid, this "oxidative stress" eats away at the body over time, eroding its ability to use energy efficiently.
And that leads to disease. Free radical damage has been linked to cancer, heart disease, diabetes, arthritis, cataracts, fatigue, hardening of the arteries and a number of neurological diseases, including Lou Gehrig's, Parkinson's and Alzheimer's.
Attack of the antioxidants
But there is a tool to fight free radicals — antioxidants. Antioxidants work by slowing down the chemical reactions that turn the normally stable oxygen molecule into a free radical.
Antioxidants such as beta carotene
and Vitamins C and E can be found in several food
products, including apples, wine, green tea, carrots and
tomatoes. But targeting free radicals with antioxidants is
easier said then done. There are about 100 antioxidants
and the way they work isn't always clearly understood.
The "Super Vitamin E" molecule spinning on
Wright's computer monitor has been designed with an
electron that has a weak bond, tempting free radicals
to steal it but not so weak that even the oxygen in
air could set it off.
In other words, he hopes the molecule will be able to
"step between the wolves and moose," in essence
"sacrificing" itself for the greater good.
"The role of the antioxidant is to be a sacrificial
weak member who dies rather than the rest of the herd. It
would react with the free radicals faster than the free
radicals can react with the DNA, proteins and
fats."
So far, the research has gone very smoothly, Wright says. Although the original grant only aimed at six molecules, he had already designed 50 models by November. Eight are already being tested on leukemia cells and the team hopes to finish on schedule within two years.
|

